 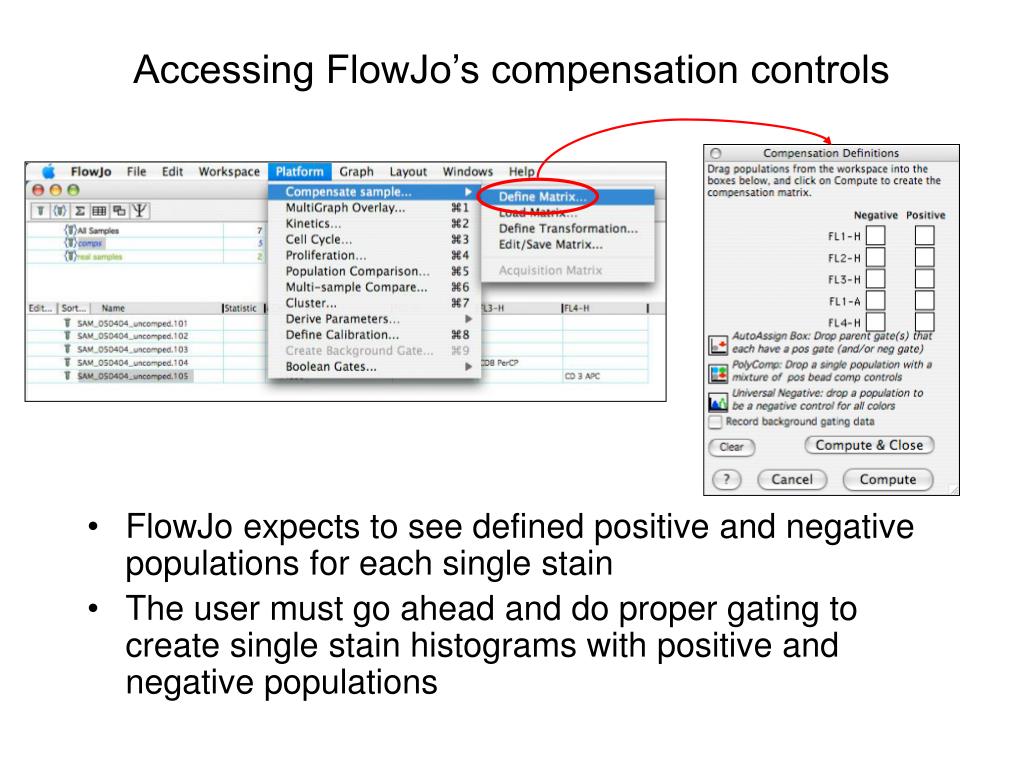
The accuracy of compensation is entirely dependent on the following good practices.įundamental Rules of Compensations Use the same reagent as was used for the fully stained sample so the spectra match identically 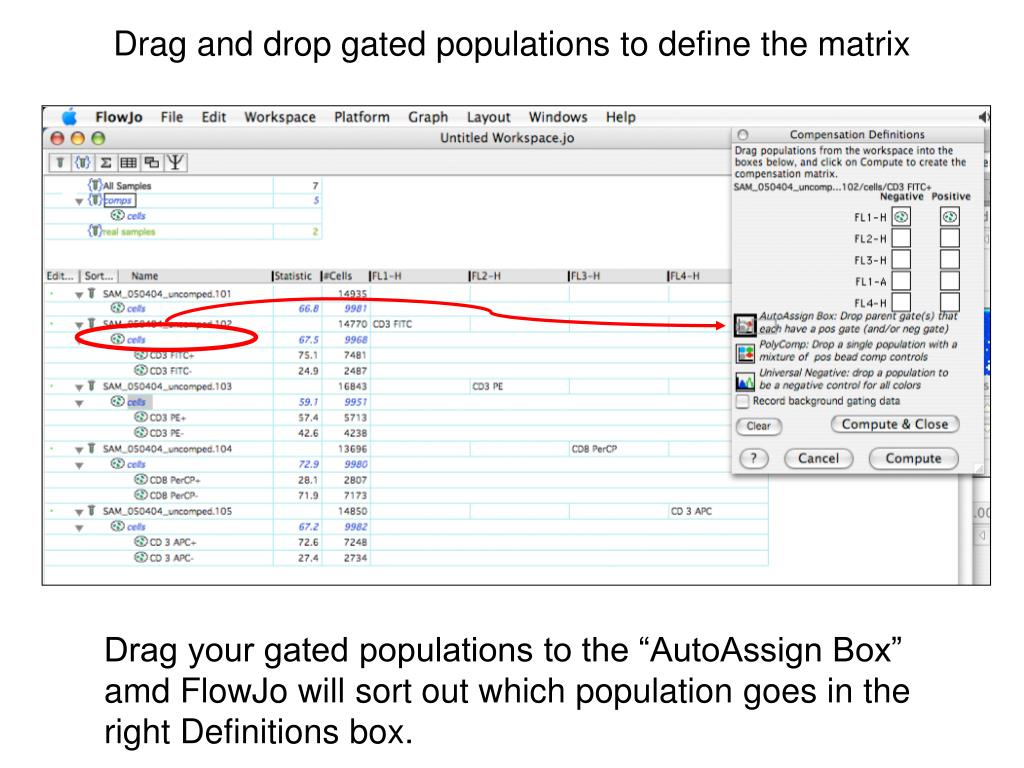
It is also possible to acquire compensation controls, compute and employ compensation post-acquisition offline in analysis using software like FCS Express or FlowJo. To measure the amount of spectral spillover in each channel, single-stained cells or antibody-binding beads, each labeled with one of the fluorescent reagents used in the panel, are separately collected at the beginning of acquisition in order to apply compensation to the subsequent samples. The visual correction of aligning a population on its axis prevents a distortion of the population that might impede accurate gating and the resolution of low abundant or double positive populations when doing bivariate gating. The amplitude and percent positive statistics of a population must always remain the same. Compensation does not change the data itself. Compensation is the mathematical correction factor that was derived from the spillover calculated using single color compensation controls and applied to both fluorophores to restore the correct alignment of the populations on all axes. When a fluorophore’s emission spills into an unintended channel, the resulting bivariate analysis plot of the fluorophore spilling (the culprit) and the channel into which it spills (the victim) will display an unusual artifact of one or both populations arcing towards the other axis rather than in a straight path along its axis. In this article, we’ll discuss considerations for effective compensation in flow cytometry. Compensation is a process to visually correct for either form of spillover, with the intention to increase the accuracy of analysis. Second is when the fluorophore is somewhat excited by other lasers besides its intended laser which is called cross-beam excitation. First is when the fluorophore has a wide emission range that spills over into a neighbor’s emission channel off the same laser, in this context called its unintended channel. Thus, there are two types of fluorescent spillover that plague flow cytometry. However, fluorophores are not discreet and the panels we employ in flow cytometry are significantly more spectrally complex than when compensation was first introduced. Flow cytometry, on the other hand, in order to increase the number of markers able to be detected and the speed of acquisition, instead began by employing 1-3 laser lines intended to excite multiple, specifically-designed tandem fluorophores with the proteins PE or APC acting as the donor fluorophore, that is the fluorophore receiving the excitation energy, and a donor fluorophore that in turn is excited by the energy of the donor fluorophore in a FRET (Förster Resonance Energy Transfer) relationship. In the early days, microscopy employed excitation sources like arc lamps, isolating specific wavelengths of light to excite single organic fluorophores as discreetly as possible. Fluorescent cellular analytical technologies allow us to “see” beyond what was historically possible with histological stains or morphological scatter profiles. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
